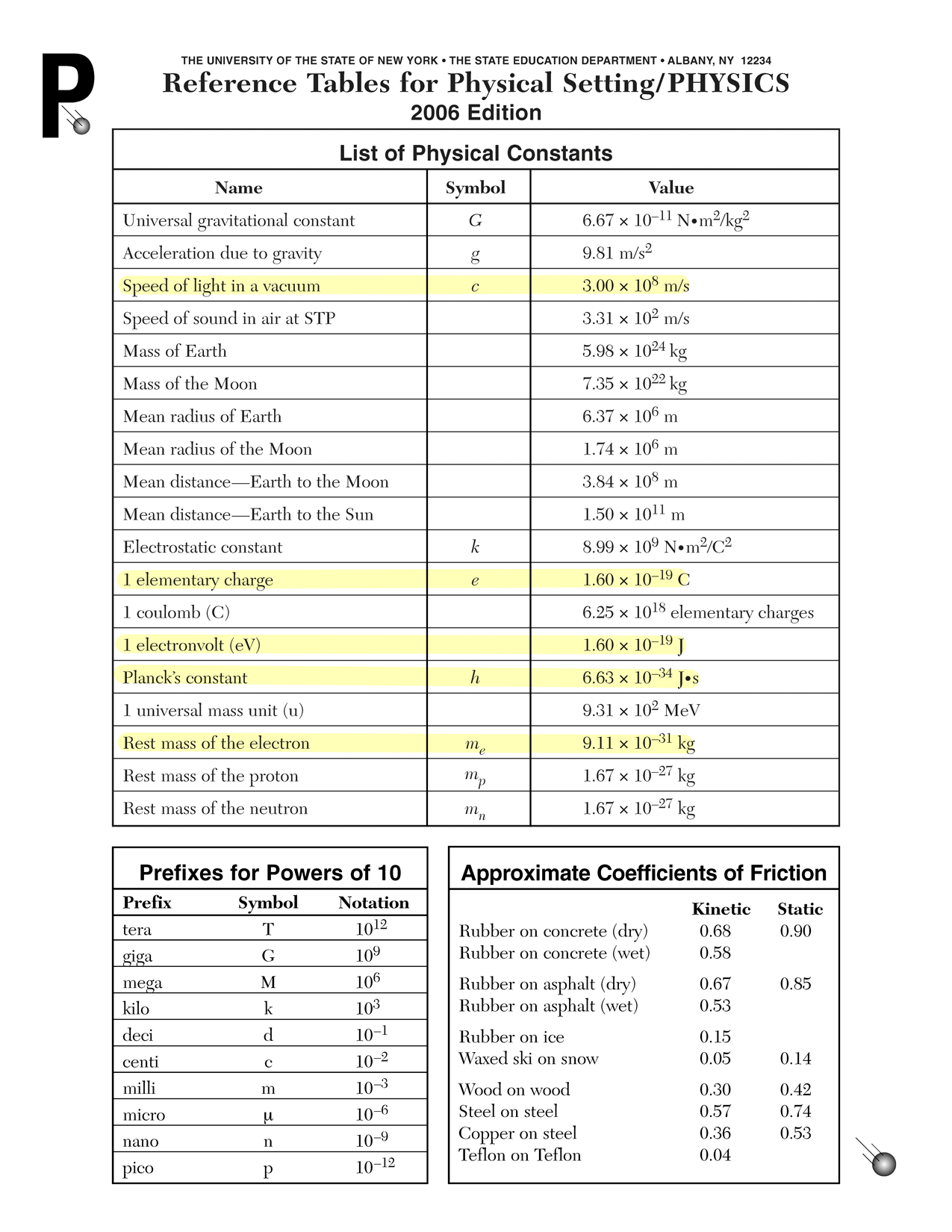
Physics A: Problem Set 25: Quantum electrodynamics
recommended reading
| High Marks: | n/a |
| Barron's Let's Review: | n/a |
| physics.info: | Quantum electrodynamics, X-rays, Antimatter |
| Wikipedia: | n/a |
| HyperPhysics: | n/a |
classwork
- A photon has just enough energy to form an electron-positron pair.
- Write this reaction out in symbolic form.
- What is the energy of this photon?
- What is its frequency?
- What is its wavelength?
- What type of electromagnetic radiation is this?
Here's the reaction in symbolic form.
00γ → 0−1e + 0+1e
A positron has the same mass as an electron. Only the charge is different. Calculate the energy equivalent to the mass of two electrons using Einstein's famous equation.
E = mc2
E = 2(9.11 × 10−31 kg)(3.00 × 108 m/s)2
E = 1.64 × 10−13 JUse Planck's equation to determine the frequency of the photon.
f = E h f = 1.64 × 10−13 J 6.63 × 10−34 J s f = 2.47 × 1020 Hz Planck's equation also comes in a version that uses wavelength instead of frequency. That's one way to solve this part of the problem.
λ = hc E λ = (6.63 × 10−34 J s)(3.00 × 108 m/s) 1.64 × 10−13 J λ = 1.21 × 10−12 m Most reference sources call this a gamma ray photon.
homework
- none